Iso 13485 internal audit checklist2/4/2024  
Requirements like those set out by ISO 13485 are strictly enforced throughout every stage of a medical device’s life-cycle, including stages after manufacturing like delivery, service, and maintenance. In the medical devices industry, quality management goes hand-in-hand with safety, and both are non-negotiables. The ISO 13485 framework also forms the basis for auditing these same organizations, for both internal and external audits. 
Perhaps the medical device industry’s most popular international standard for quality management, ISO 13485 provides a framework for manufacturers to implement the Medical Device Directives while simultaneously demonstrating a commitment to the quality and safety guidelines of medical devices.Īs of writing, the most recent version of the standard is ISO 13485:2016.īasically, ISO 13485 is like a quality management system for organizations involved in design, production, installation, and servicing of medical devices, with some other important requirements for good measure. Simply put, ISO 13485 is a set of requirements defined by The International Organization for Standardization, designed to be used by medical device manufacturers as a form of quality management system. In this article, I’ll break down the ISO 13485 standard, from a basic introduction to suggestions and resources for implementing it in your business or organization. Often, these kinds of requirements take the form of the ISO 13485 standard for medical device manufacturers. At some point soon, the current internal systems will not be able to hold back the deluge, and companies will be faced with a stark decision-consistently improve or perish” – Erik Myhrberg and Joseph Raciti, Practical Field Guide for ISO 13485 “On a global scale, we are all being asked to do more with less-and for less. That’s just the tip of the iceberg more severe penalties extend to include government bodies compelling you to dissolve your company, and ultimately the endangerment of the lives and well-being of individuals your organization is servicing. Failure to comply with several licencing regulations issued by the California Department of Insurance landed them a $7million fine. However, the struggle to turn a profit pales in comparison to some of the harsher consequences of failing to comply with certain regulatory requirements. CAPA also calls for measures to help mitigate and prevent future occurrences of the same quality issues.In today’s business world, owners are constantly grappling with concerns and surmounting obstacles, the least of which is actually staying afloat financially in what can be an unforgiving economy. It’s not enough to correct nonconformances and other quality issues. Evaluate steps taken to resolve nonconformances.CAPA implementation should be formally documented and its records maintained. Implement correction of nonconformances and their causesĬorrective action and preventive action (CAPA) is a crucial aspect of ISO audit procedures.The documentation aspect of an internal audit program should include policies, procedures, instructions, and records of audits that have been performed. Keep a record of the audit plan and performance.Most companies typically perform it on an annual or semi-annual basis, though the frequency should depend on the organization’s needs. ISO standards require regular internal audits. Perform internal audits at planned intervals.ISO audit requirements call for a formal audit process, which implies the need for a documented plan that can be shared throughout the organization. Plan the organization’s internal audit program.A company’s quality policy should include a formal internal audit program with documentation and implementation components. 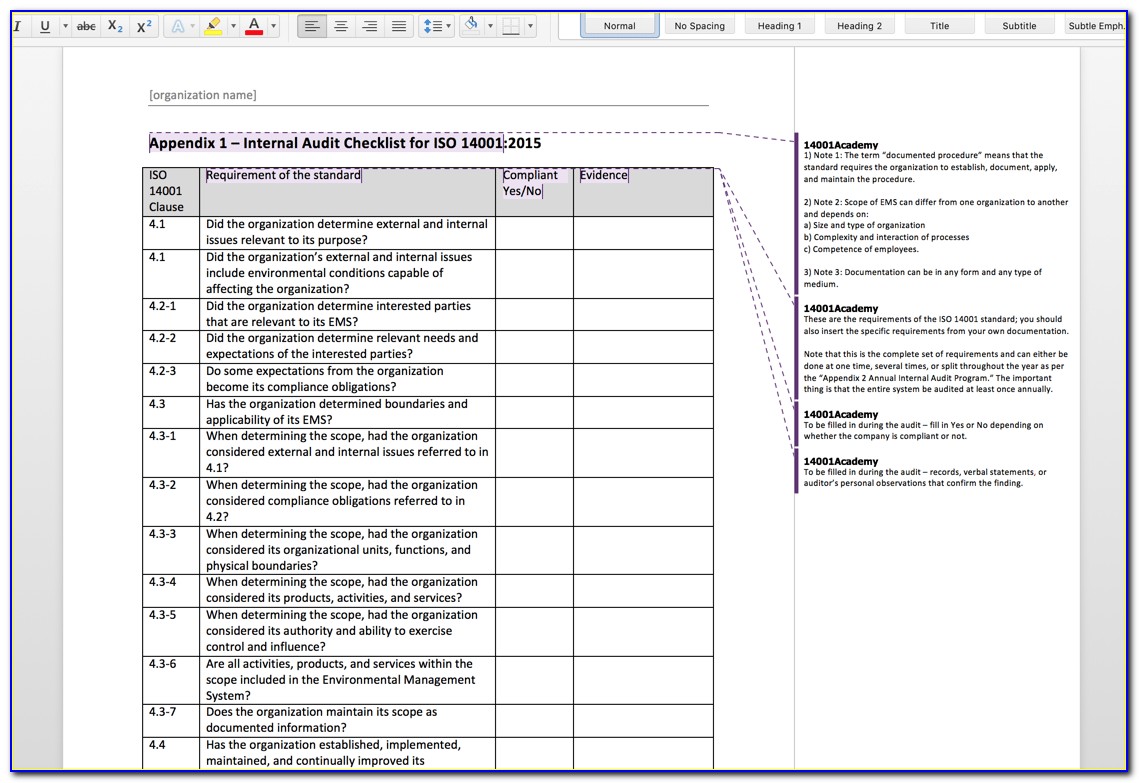
An internal audit process is meant to assess the strength of an organization’s quality processes and uncover any weaknesses.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
